Original source: Medical Physics, Radiation Oncology & Cancer
This article is an editorial summary and interpretation of that content. The ideas belong to the original authors; the selection and writing are by Streamed.News.
This video from Medical Physics, Radiation Oncology & Cancer covered a lot of ground. 6 segments stood out as worth your time. Everything below links directly to the timestamp in the original video.
Mastering the Normal Tissue Objective (NTO) in Eclipse can significantly enhance the precision of your radiation therapy plans. Learn how adjusting NTO parameters like distance, start/end dose, and fall-off slope directly shapes the dose distribution around your targets.
Eclipse NTO Parameters Offer Precise Dose Fall-Off Control
Radiation therapy planners can precisely control dose fall-off in Eclipse by adjusting Normal Tissue Objective (NTO) parameters, which include the distance from the target border, the start dose, the end dose, and the fall-off slope. The key is to visually monitor the NTO graph, which provides immediate feedback on the optimizer's intended dose distribution. While various parameter combinations can achieve similar results, a fall-off slope between 0 and 10 allows for shaping the dose curve, with 10 representing an ideal, sharp dose drop.
This level of granular control is crucial for tailoring radiation plans, especially in complex cases like VMAT chest wall treatments. For instance, setting a distance of one centimeter from the target border with a 70% start dose and 20% end dose can strategically allow a higher dose to spill outside the Planning Target Volume (PTV) while sparing critical structures like the lungs. Understanding these parameters enables clinicians to optimize treatment delivery, ensuring effective tumor targeting with minimal impact on surrounding healthy tissues.
"The main thing is to look at that little graph on the right and that's going to show you what you want or what you are telling the optimizer to make the dose fall off look like."
Split Field Technique Improves Head and Neck Radiation Planning
A specialized split field technique in radiation therapy, designed for patients with long node chains, significantly enhances plan quality and spares critical structures in the head and neck. This method employs four radiation fields with collimators rotated 90 degrees and two fields with fixed jaws set at approximately zero degrees. By pre-defining jaw coordinates before optimization, the system prevents them from opening beyond a set limit, eliminating open segments between node chains and allowing the optimizer to focus more effectively on the Planning Target Volume (PTV).
This approach particularly benefits head and neck treatments by better protecting sensitive organs such as the larynx, esophagus, and spinal cord. While some may express concerns about increased integral dose or monitor units (MU), clinical experience suggests that this technique typically uses around 150 MU per field, adds only two to three minutes to treatment time, and often results in lower integral dose due to more precise targeting. Physicians should consider this method for most patients to achieve notably improved plan quality.
"I think the biggest benefit of this is that you're not going to have open segments between the two node chains, and that kind of helps especially on head and necks when you have things like larynx, esophagus, cord, all those things in the middle here, it really helps spare those structures."
Weight Loss During Head and Neck Treatment Prompts Dose Re-Evaluation
Managing significant weight loss during head and neck radiation therapy, a common occurrence around fraction 25 of a 35-fraction regimen, requires careful re-evaluation of patient dosimetry to prevent unintended dose increases. A general rule of thumb indicates that a one-centimeter change in tissue depth can alter the dose from a 6MV beam by approximately 3.5% at clinically relevant depths. Since weight loss is rarely uniform, a patient experiencing five millimeters of lateral shrinkage and one centimeter anterior tissue loss might see a roughly 2% increase in overall dose.
Clinicians must assess whether normal structures have shifted into high-dose regions or if the tumor has moved outside the Planning Target Volume (PTV). If such shifts occur, a re-scan (re-CT) or re-planning is necessary. If only shrinkage is observed without significant anatomical shifts, a cone beam CT (CBCT) can be used to define a new body contour and recalculate the existing plan in Eclipse, leveraging features like Boolean operators and the Shift F5 command, to obtain a more accurate current dose distribution without requiring a full re-plan.
"If normal structures start to move so like if a patient lost weight in their shoulders... that would be a time when you would definitely need to re-scan."
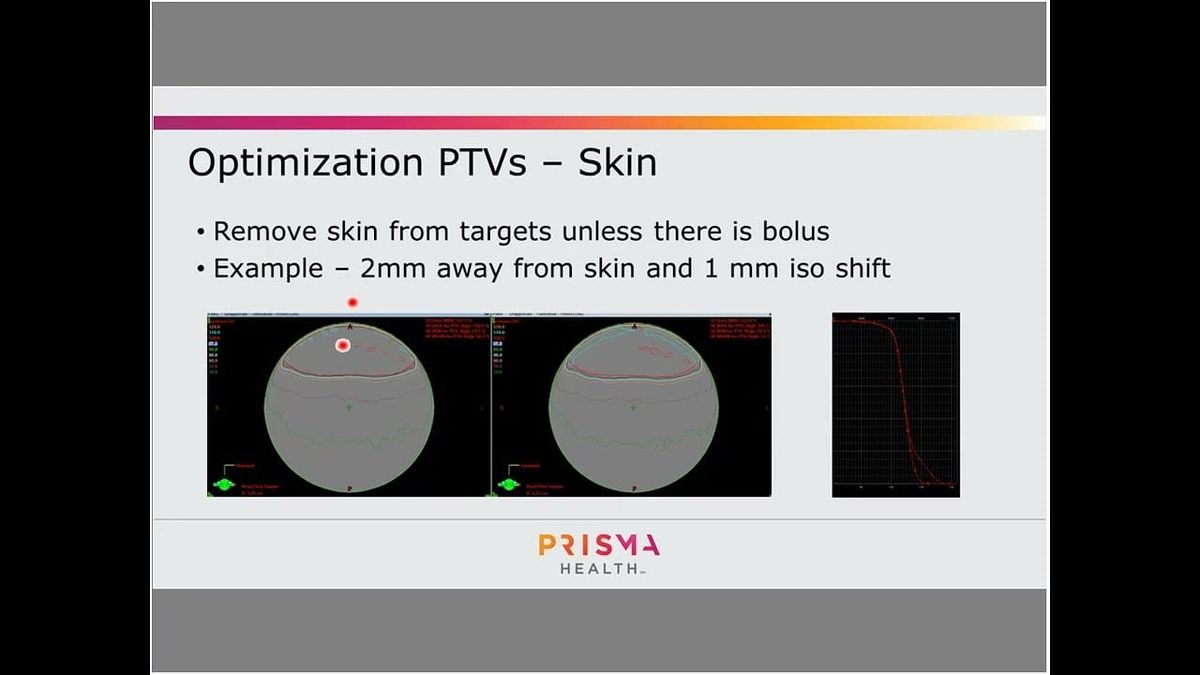
Avoid Skin in PTVs to Prevent Hot Spots, Improve Dose Distribution
To prevent unintended hot spots and improve dose distribution in radiation therapy, it is crucial to exclude the skin from Planning Target Volumes (PTVs) or use a bolus. Radiation optimizers inherently struggle to accurately model dose in superficial tissues because photon beams require several millimeters to centimeters for dose build-up, meaning the maximum dose occurs below the surface. Consequently, optimizers often compensate by introducing excessive tangential modulation, which appears effective on paper but becomes highly susceptible to patient shifts during treatment, potentially leading to localized high-dose areas on the skin.
For example, even a one-millimeter patient shift in a plan with a two-millimeter PTV-to-skin distance can cause 20% of the PTV to receive significantly higher doses, escalating to 50% if optimizing directly to the skin. To mitigate this risk, clinicians should either apply a bolus when skin treatment is desired or subtract at least five millimeters from the skin when defining PTVs. Eclipse's position uncertainty tool can also simulate the impact of patient shifts, allowing planners to assess the robustness of their plans and avoid tangential dose accumulation.
"On paper it might look fine but in reality you have a very high likelihood of delivering a lot of dose to a patient if they move any."
Advanced Eclipse Optimization Boosts Plan Quality and Accuracy
Radiation therapy planners can significantly enhance plan quality and optimizer accuracy in Eclipse by implementing several advanced optimization techniques. One method involves applying mean dose constraints to optimization structures, such as a parotid gland with the Planning Target Volume (PTV) subtracted, which helps protect critical organs without compromising target coverage. Additionally, setting high priorities, specifically a value of 1000, for certain dose constraints—like a cord dose of 35 Gray or a parotid mean dose of 20 Gray—effectively 'holds' these objectives, allowing the optimizer to focus on refining other normal tissue sparing without violating established limits.
This high-priority technique also works for PTV coverage, ensuring specific percentages are met while other objectives are pursued. Furthermore, performing intermediate dose calculations is particularly beneficial for structures adjacent to air cavities, such as sinuses or the nasopharynx. These additional calculations improve the agreement between optimized and final dose distributions, preventing significant discrepancies in the dose-volume histograms (DVHs) that can occur in regions with heterogeneous tissue densities, thereby leading to more reliable and accurate plans.
"I found that the more intermediate dose calcs you do, you actually get better agreement."
Optimization PTVs Control Head and Neck Dose Spillage
In head and neck radiation planning, optimization Planning Target Volumes (PTVs) are crucial for precisely controlling dose fall-off and minimizing spillage from higher dose PTVs into adjacent lower dose PTVs. This technique prevents healthy tissue or lower-risk target volumes, such as a PTV 56, from inadvertently receiving excessively high doses like 65 Gray. A recommended method for creating these structures involves expanding a higher dose PTV by one millimeter for every one percent dose difference, often resulting in a one-centimeter expansion, then subtracting it from the next lower dose PTV to create an optimization PTV (e.g., PTV63 - Expanded PTV70).
Within Eclipse, specific constraints are applied to these optimization PTVs to ensure tight dose control. It is vital to set a lower objective on the actual PTV to guarantee full coverage and an upper objective near the higher dose PTV's level to manage dose fall-off effectively. If the optimization PTV expansion is too small (e.g., one to two millimeters), it can compromise coverage of the higher dose targets, leading to unintended dose squeezing and fall-off between structures. Properly constructed optimization PTVs allow for efficient and accurate dose planning.
"The goal of the optimization PTVs is to keep those doses from spilling into the to the lower PTVs."
Also mentioned in this video
- Head and neck treatments are considered challenging due to a high number of… (2:00)
- Additional optimization structures include rings, a posterior void to spare the… (13:53)
- RapidPlan is an excellent starting point for optimization due to its estimate… (18:22)
- Other optimization strategies include pausing the optimizer to adjust… (23:18)
- The cost function tool in Eclipse provides insight into how the optimizer… (28:44)
- Decisions on sparing structures versus covering targets are… (30:57)
- Re-treatment cases involving doses exceeding 100 Gray require careful physician… (37:35)
- Effective head and neck planning involves a wide range of treatment techniques,… (40:17)
Summarised from Medical Physics, Radiation Oncology & Cancer · 41:51. All credit belongs to the original creators. Streamed.News summarises publicly available video content.
Streamed.News
This publication is generated automatically from YouTube.
Convert your full video library into a digital newspaper.
Get this for your newsroom →